[1] Wu, Z., & McGoogan, J. M. (2020). Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA, 323(13), 1239–1242. https://doi.org/10.1001/jama.2020.2648
[2] Richardson, S., Hirsch, J. S., Narasimhan, M., Crawford, J. M., McGinn, T., Davidson, K. W., the Northwell COVID-19 Research Consortium, Barnaby, D. P., Becker, L. B., Chelico, J. D., Cohen, S. L., Cookingham, J., Coppa, K., Diefenbach, M. A., Dominello, A. J., Duer-Hefele, J., Falzon, L., Gitlin, J., Hajizadeh, N., Harvin, T. G., … Zanos, T. P. (2020). Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA, 323(20), 2052–2059. https://doi.org/10.1001/jama.2020.6775
[3] https://www.cdc.gov/aging/covid19-guidance.html
[4] Santesmasses, D., Castro, J. P., Zenin, A. A., Shindyapina, A. V., Gerashchenko, M. V., Zhang, B., Kerepesi, C., Yim, S. H., Fedichev, P. O., & Gladyshev, V. N. (2020). COVID-19 is an emergent disease of aging. Aging cell, 19(10), e13230. https://doi.org/10.1111/acel.13230
[5] Chow, R. D., Majety, M., & Chen, S. (2021). The aging transcriptome and cellular landscape of the human lung in relation to SARS-CoV-2. Nature communications, 12(1), 4. https://doi.org/10.1038/s41467-020-20323-9
[6] https://www.geron.org/press-room/press-releases/2020-press-releases
[7] Sanchez-Vazquez, R., Guío-Carrión, A., Zapatero-Gaviria, A., Martínez, P., & Blasco, M. A. (2021). Shorter telomere lengths in patients with severe COVID-19 disease. Aging, 13(1), 1–15. https://doi.org/10.18632/aging.202463
[8] Sepe, S., Rossiello, F., Cancila, V., Iannelli, F., Matti, V., Cicio, G., Cabrini, M., Marinelli, E., Alabi, B. R., di Lillo, A., Di Napoli, A., Shay, J. W., Tripodo, C., & d'Adda di Fagagna, F. (2022). DNA damage response at telomeres boosts the transcription of SARS-CoV-2 receptor ACE2 during aging. EMBO reports, 23(2), e53658. https://doi.org/10.15252/embr.202153658
[9] Lee, S., Yu, Y., Trimpert, J. et al. (2021). Virus-induced senescence is driver and therapeutic target in COVID-19. Nature. https://doi.org/10.1038/s41586-021-03995-1
[10] Cox, L. S., Bellantuono, I., Lord, J. M., Sapey, E., Mannick, J. B., Partridge, L., Gordon, A. L., Steves, C. J., & Witham, M. D. (2020). Tackling immunosenescence to improve COVID-19 outcomes and vaccine response in older adults. The Lancet. Healthy longevity, 1(2), e55–e57. https://doi.org/10.1016/S2666-7568(20)30011-8
[11] Chen, Y., Klein, S. L., Garibaldi, B. T., Li, H., Wu, C., Osevala, N. M., Li, T., Margolick, J. B., Pawelec, G., & Leng, S. X. (2021). Aging in COVID-19: Vulnerability, immunity and intervention. Ageing research reviews, 65, 101205. https://doi.org/10.1016/j.arr.2020.101205
[12] Andrews, M., Soto, N., & Arredondo, M. (2012). Efecto de metformina sobre la expresión del factor de necrosis tumoral-α, los receptores Toll-like 2/4 y la PCR ultra sensible en sujetos obesos con diabetes tipo 2 [Effect of metformin on the expression of tumor necrosis factor-α, Toll like receptors 2/4 and C reactive protein in obese type-2 diabetic patients]. Revista medica de Chile, 140(11), 1377–1382. https://doi.org/10.4067/S0034-98872012001100001
[13] Zahra, M. H., Afify, S. M., Hassan, G., Nawara, H. M., Kumon, K., Seno, A., & Seno, M. (2021). Metformin suppresses self-renewal and stemness of cancer stem cell models derived from pluripotent stem cells. Cell biochemistry and function, 39(7), 896–907. https://doi.org/10.1002/cbf.3661
[14] Dahabiyeh, L. A., Mujammami, M., Arafat, T., Benabdelkamel, H., Alfadda, A. A., & Abdel Rahman, A. M. (2021). A Metabolic Pattern in Healthy Subjects Given a Single Dose of Metformin: A Metabolomics Approach. Frontiers in pharmacology, 12, 705932. https://doi.org/10.3389/fphar.2021.705932
[15] Novelle, M. G., Ali, A., Diéguez, C., Bernier, M., & de Cabo, R. (2016). Metformin: A Hopeful Promise in Aging Research. Cold Spring Harbor perspectives in medicine, 6(3), a025932. https://doi.org/10.1101/cshperspect.a025932
[16] Crouse, A. B., Grimes, T., Li, P., Might, M., Ovalle, F., & Shalev, A. (2021). Metformin Use Is Associated With Reduced Mortality in a Diverse Population With COVID-19 and Diabetes. Frontiers in endocrinology, 11, 600439. https://doi.org/10.3389/fendo.2020.600439
[17] Cariou, B., Hadjadj, S., Wargny, M., Pichelin, M., Al-Salameh, A., Allix, I., Amadou, C., Arnault, G., Baudoux, F., Bauduceau, B., Borot, S., Bourgeon-Ghittori, M., Bourron, O., Boutoille, D., Cazenave-Roblot, F., Chaumeil, C., Cosson, E., Coudol, S., Darmon, P., Disse, E., … CORONADO investigators (2020). Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia, 63(8), 1500–1515. https://doi.org/10.1007/s00125-020-05180-x
[18] Bramante, C. T., Ingraham, N. E., Murray, T. A., Marmor, S., Hovertsen, S., Gronski, J., McNeil, C., Feng, R., Guzman, G., Abdelwahab, N., King, S., Tamariz, L., Meehan, T., Pendleton, K. M., Benson, B., Vojta, D., & Tignanelli, C. J. (2021). Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis. The Lancet. Healthy longevity, 2(1), e34–e41. https://doi.org/10.1016/S2666-7568(20)30033-7
[19] Lukito, A. A., Pranata, R., Henrina, J., Lim, M. A., Lawrensia, S., & Suastika, K. (2020). The Effect of Metformin Consumption on Mortality in Hospitalized COVID-19 patients: a systematic review and meta-analysis. Diabetes & metabolic syndrome, 14(6), 2177–2183. https://doi.org/10.1016/j.dsx.2020.11.006
[20] Luo, P., Qiu, L., Liu, Y., Liu, X. L., Zheng, J. L., Xue, H. Y., Liu, W. H., Liu, D., & Li, J. (2020). Metformin Treatment Was Associated with Decreased Mortality in COVID-19 Patients with Diabetes in a Retrospective Analysis. The American journal of tropical medicine and hygiene, 103(1), 69–72. https://doi.org/10.4269/ajtmh.20-0375
[21] Sharma, S., Ray, A., & Sadasivam, B. (2020). Metformin in COVID-19: A possible role beyond diabetes. Diabetes research and clinical practice, 164, 108183. https://doi.org/10.1016/j.diabres.2020.108183
[22] Esam Z. (2020). A proposed mechanism for the possible therapeutic potential of Metformin in COVID-19. Diabetes research and clinical practice, 167, 108282. https://doi.org/10.1016/j.diabres.2020.108282
[23] Ursini, F., Russo, E., Pellino, G., D'Angelo, S., Chiaravalloti, A., De Sarro, G., Manfredini, R., & De Giorgio, R. (2018). Metformin and Autoimmunity: A "New Deal" of an Old Drug. Frontiers in immunology, 9, 1236. https://doi.org/10.3389/fimmu.2018.01236
[24] Afshari, K., Dehdashtian, A., Haddadi, N. S., Haj-Mirzaian, A., Iranmehr, A., Ebrahimi, M. A., Tavangar, S. M., Faghir-Ghanesefat, H., Mohammadi, F., Rahimi, N., Javidan, A. N., & Dehpour, A. R. (2018). Anti-inflammatory effects of Metformin improve the neuropathic pain and locomotor activity in spinal cord injured rats: introduction of an alternative therapy. Spinal cord, 56(11), 1032–1041. https://doi.org/10.1038/s41393-018-0168-x
[25] Bendib, I., de Chaisemartin, L., Granger, V., Schlemmer, F., Maitre, B., Hüe, S., Surenaud, M., Beldi-Ferchiou, A., Carteaux, G., Razazi, K., Chollet-Martin, S., Mekontso Dessap, A., & de Prost, N. (2019). Neutrophil Extracellular Traps Are Elevated in Patients with Pneumonia-related Acute Respiratory Distress Syndrome. Anesthesiology, 130(4), 581–591. https://doi.org/10.1097/ALN.0000000000002619
[26] Zuo, Y., Yalavarthi, S., Shi, H., Gockman, K., Zuo, M., Madison, J. A., Blair, C., Weber, A., Barnes, B. J., Egeblad, M., Woods, R. J., Kanthi, Y., & Knight, J. S. (2020). Neutrophil extracellular traps in COVID-19. JCI insight, 5(11), e138999. https://doi.org/10.1172/jci.insight.138999
[27] Thoppil, H., & Riabowol, K. (2020). Senolytics: A Translational Bridge Between Cellular Senescence and Organismal Aging. Frontiers in cell and developmental biology, 7, 367. https://doi.org/10.3389/fcell.2019.00367
[28] Camell, C. D., Yousefzadeh, M. J., Zhu, Y., Prata, L., Huggins, M. A., Pierson, M., Zhang, L., O'Kelly, R. D., Pirtskhalava, T., Xun, P., Ejima, K., Xue, A., Tripathi, U., Espindola-Netto, J. M., Giorgadze, N., Atkinson, E. J., Inman, C. L., Johnson, K. O., Cholensky, S. H., Carlson, T. W., Robbins, P. D. (2021). Senolytics reduce coronavirus-related mortality in old mice. Science (New York, N.Y.), 373(6552), eabe4832. https://doi.org/10.1126/science.abe4832
[29] https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3677428
[30] Altay, O., Arif, M., Li, X., Yang, H., Aydın, M., Alkurt, G., Kim, W., Akyol, D., Zhang, C., Dinler‐Doganay, G., Turkez, H., Shoaie, S., Nielsen, J., Borén, J., Olmuscelik, O., Doganay, L., Uhlén, M., & Mardinoglu, A. (2021). Combined Metabolic Activators Accelerates Recovery in Mild‐to‐Moderate COVID‐19. Advanced Science, 8(17), 2101222. https://doi.org/10.1002/advs.202101222
[31] Zheng, M., Schultz, M. B., & Sinclair, D. A. (2022). NAD+ in COVID-19 and viral infections. Trends in immunology, 43(4), 283–295. https://doi.org/10.1016/j.it.2022.02.001
[32] Jiang, Y., Deng, Y., Pang, H., Ma, T., Ye, Q., Chen, Q., Chen, H., Hu, Z., Qin, C. F., & Xu, Z. (2022). Treatment of SARS-CoV-2-induced pneumonia with NAD+ and NMN in two mouse models. Cell discovery, 8(1), 38. https://doi.org/10.1038/s41421-022-00409-y
[33] https://www.technologyreview.com/2022/05/17/1052334/anti-aging-drugs-treat-covid-19/
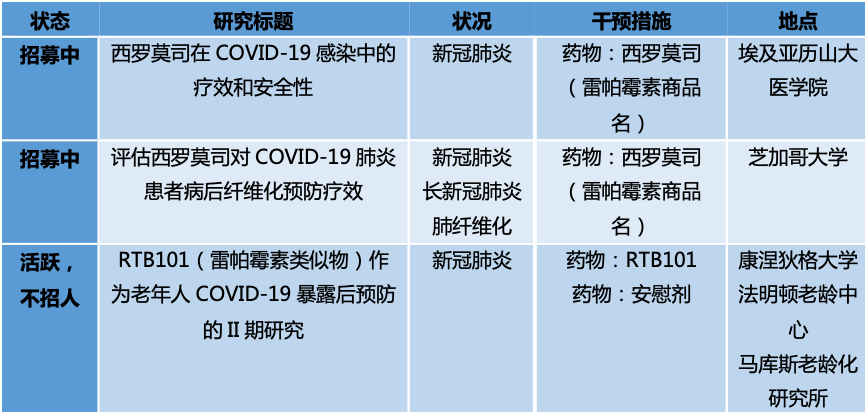
[34] Patocka, J., Kuca, K., Oleksak, P., Nepovimova, E., Valis, M., Novotny, M., & Klimova, B. (2021). Rapamycin: Drug Repurposing in SARS-CoV-2 Infection. Pharmaceuticals (Basel, Switzerland), 14(3), 217. https://doi.org/10.3390/ph14030217
[35] Bischof, E., Siow, R. C., Zhavoronkov, A., & Kaeberlein, M. (2021). The potential of rapalogs to enhance resilience against SARS-CoV-2 infection and reduce the severity of COVID-19. The Lancet. Healthy longevity, 2(2), e105–e111. https://doi.org/10.1016/S2666-7568(20)30068-4
[36] Cawthon, R. M., Smith, K. R., O'Brien, E., Sivatchenko, A., & Kerber, R. A. (2003). Association between telomere length in blood and mortality in people aged 60 years or older. Lancet (London, England), 361(9355), 393–395. https://doi.org/10.1016/S0140-6736(03)12384-7
[37] Brouilette, S. W., Moore, J. S., McMahon, A. D., Thompson, J. R., Ford, I., Shepherd, J., Packard, C. J., Samani, N. J., & West of Scotland Coronary Prevention Study Group (2007). Telomere length, risk of coronary heart disease, and statin treatment in the West of Scotland Primary Prevention Study: a nested case-control study. Lancet (London, England), 369(9556), 107–114. https://doi.org/10.1016/S0140-6736(07)60071-3
[38] Hashimoto, M., & Akita, H. (2002). Cerivastatin, a hydroxymethylglutaryl coenzyme a reductase inhibitor, improves endothelial function in elderly diabetic patients within 3 days. Circulation, 105(4), E30–E31.
[39] Zhang, X. J., Qin, J. J., Cheng, X., Shen, L., Zhao, Y. C., Yuan, Y., Lei, F., Chen, M. M., Yang, H., Bai, L., Song, X., Lin, L., Xia, M., Zhou, F., Zhou, J., She, Z. G., Zhu, L., Ma, X., Xu, Q., Ye, P., … Li, H. (2020). In-Hospital Use of Statins Is Associated with a Reduced Risk of Mortality among Individuals with COVID-19. Cell metabolism, 32(2), 176–187.e4. https://doi.org/10.1016/j.cmet.2020.06.015
[40] Iacobucci G. (2021). Covid-19: People who take statins may be less likely to die, research suggests. BMJ (Clinical research ed.), 375, n2536. https://doi.org/10.1136/bmj.n2536
[41] https://heal-covid.net/
[42] https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/index.html
[43] https://www.nia.nih.gov/research/blog/2022/04/nia-budget-and-pay-lines-update-springing-forward
[44] https://www.science.org/content/article/biden-s-first-budget-request-goes-big-science
[45] https://grants.nih.gov/grants/guide/RFA-files/RFA-AG-23-016.html