[1]Lopez-Leon, S., Wegman-Ostrosky, T., Perelman, C., Sepulveda, R., Rebolledo, P. A., Cuapio, A., & Villapol, S. (2021). More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. Scientific reports, 11(1), 16144. https://doi.org/10.1038/s41598-021-95565-8
[2]Xu, E., Xie, Y., & Al-Aly, Z. (2022). Long-term neurologic outcomes of COVID-19. Nature medicine, 28(11), 2406–2415. https://doi.org/10.1038/s41591-022-02001-z
[3]Aluganti Narasimhulu, C., & Singla, D. K. (2022). Mechanisms of COVID-19 pathogenesis in diabetes. American journal of physiology. Heart and circulatory physiology, 323(3), H403–H420. https://doi.org/10.1152/ajpheart.00204.2022
[4]https://www.nature.com/articles/d41586-022-04502-w
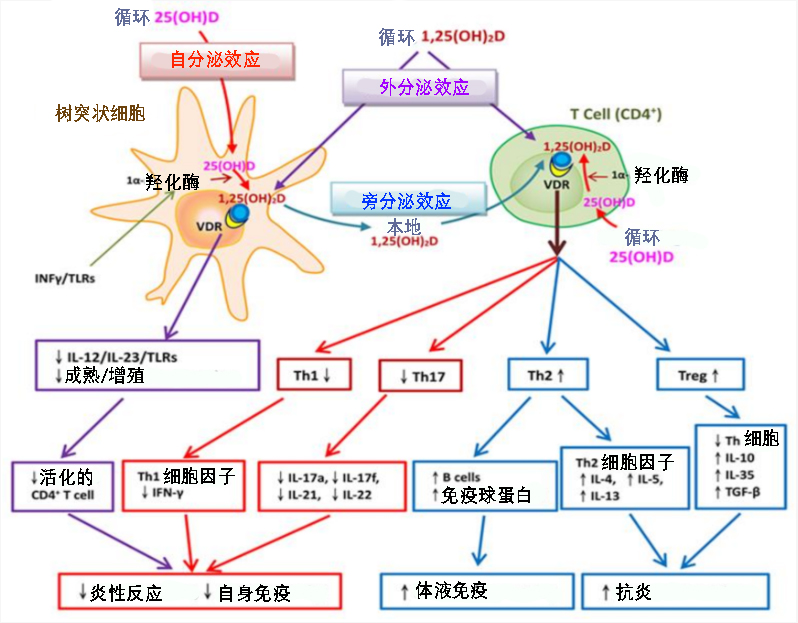
[5]Teshome, A., Adane, A., Girma, B., & Mekonnen, Z. A. (2021). The Impact of Vitamin D Level on COVID-19 Infection: Systematic Review and Meta-Analysis. Frontiers in public health, 9, 624559. https://doi.org/10.3389/fpubh.2021.624559
[6]Mishra, P., Parveen, R., Bajpai, R., & Agarwal, N. (2022). Vitamin D Deficiency and Comorbidities as Risk Factors of COVID-19 Infection: A Systematic Review and Meta-analysis. Journal of preventive medicine and public health = Yebang Uihakhoe chi, 55(4), 321–333. https://doi.org/10.3961/jpmph.21.640
[7]Corrao, S., Mallaci Bocchio, R., Lo Monaco, M., Natoli, G., Cavezzi, A., Troiani, E., & Argano, C. (2021). Does Evidence Exist to Blunt Inflammatory Response by Nutraceutical Supplementation during COVID-19 Pandemic? An Overview of Systematic Reviews of Vitamin D, Vitamin C, Melatonin, and Zinc. Nutrients, 13(4), 1261. https://doi.org/10.3390/nu13041261
[8]Jolliffe, D. A., Camargo, C. A., Jr, Sluyter, J. D., Aglipay, M., Aloia, J. F., Ganmaa, D., Bergman, P., Bischoff-Ferrari, H. A., Borzutzky, A., Damsgaard, C. T., Dubnov-Raz, G., Esposito, S., Gilham, C., Ginde, A. A., Golan-Tripto, I., Goodall, E. C., Grant, C. C., Griffiths, C. J., Hibbs, A. M., Janssens, W., … Martineau, A. R. (2021). Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials. The lancet. Diabetes & endocrinology, 9(5), 276–292. https://doi.org/10.1016/S2213-8587(21)00051-6
[9]Mitchell F. (2020). Vitamin-D and COVID-19: do deficient risk a poorer outcome?. The lancet. Diabetes & endocrinology, 8(7), 570. https://doi.org/10.1016/S2213-8587(20)30183-2
[10]Peng, M. Y., Liu, W. C., Zheng, J. Q., Lu, C. L., Hou, Y. C., Zheng, C. M., Song, J. Y., Lu, K. C., & Chao, Y. C. (2021). Immunological Aspects of SARS-CoV-2 Infection and the Putative Beneficial Role of Vitamin-D. International journal of molecular sciences, 22(10), 5251. https://doi.org/10.3390/ijms22105251
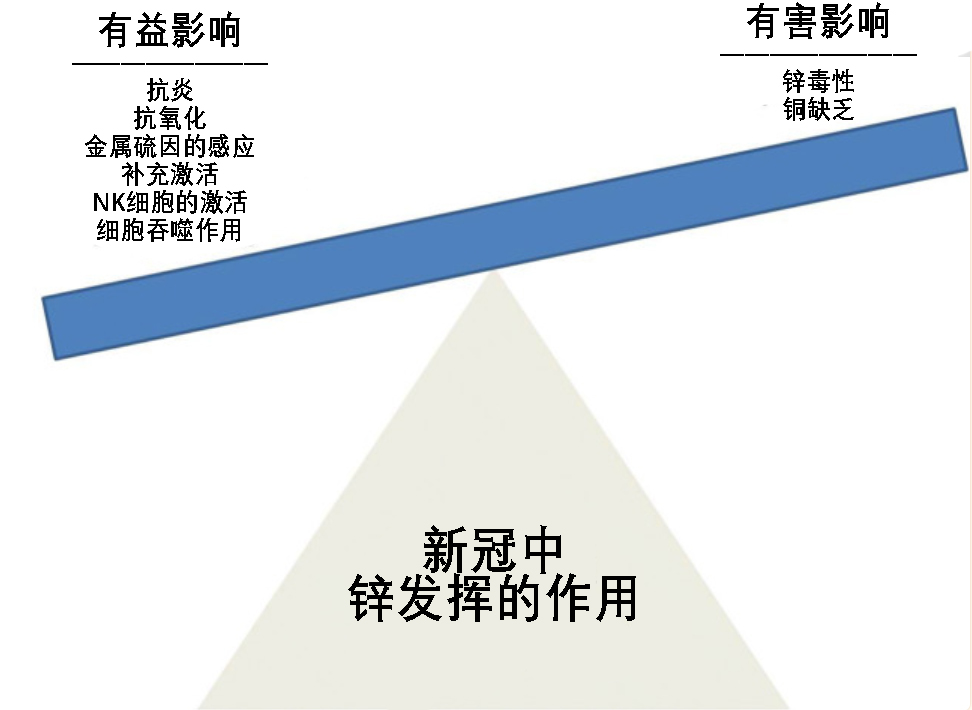
[11]Hunter, J., Arentz, S., Goldenberg, J., Yang, G., Beardsley, J., Myers, S. P., Mertz, D., & Leeder, S. (2021). Zinc for the prevention or treatment of acute viral respiratory tract infections in adults: a rapid systematic review and meta-analysis of randomised controlled trials. BMJ open, 11(11), e047474. https://doi.org/10.1136/bmjopen-2020-047474
[12]Abioye, A. I., Bromage, S., & Fawzi, W. (2021). Effect of micronutrient supplements on influenza and other respiratory tract infections among adults: a systematic review and meta-analysis. BMJ global health, 6(1), e003176. https://doi.org/10.1136/bmjgh-2020-003176
[13]Tabatabaeizadeh S. A. (2022). Zinc supplementation and COVID-19 mortality: a meta-analysis. European journal of medical research, 27(1), 70. https://doi.org/10.1186/s40001-022-00694-z
[14]Vlieg-Boerstra, B., de Jong, N., Meyer, R., Agostoni, C., De Cosmi, V., Grimshaw, K., Milani, G. P., Muraro, A., Oude Elberink, H., Pali-Schöll, I., Roduit, C., Sasaki, M., Skypala, I., Sokolowska, M., van Splunter, M., Untersmayr, E., Venter, C., O'Mahony, L., & Nwaru, B. I. (2022). Nutrient supplementation for prevention of viral respiratory tract infections in healthy subjects: A systematic review and meta-analysis. Allergy, 77(5), 1373–1388. https://doi.org/10.1111/all.15136
[15]Hunter, J., Arentz, S., Goldenberg, J., Yang, G., Beardsley, J., Myers, S. P., Mertz, D., & Leeder, S. (2021). Zinc for the prevention or treatment of acute viral respiratory tract infections in adults: a rapid systematic review and meta-analysis of randomised controlled trials. BMJ open, 11(11), e047474. https://doi.org/10.1136/bmjopen-2020-047474
[16]Pal, A., Squitti, R., Picozza, M., Pawar, A., Rongioletti, M., Dutta, A. K., Sahoo, S., Goswami, K., Sharma, P., & Prasad, R. (2021). Zinc and COVID-19: Basis of Current Clinical Trials. Biological trace element research, 199(8), 2882–2892. https://doi.org/10.1007/s12011-020-02437-9
[17]Gutiérrez, S., Svahn, S. L., & Johansson, M. E. (2019). Effects of Omega-3 Fatty Acids on Immune Cells. International journal of molecular sciences, 20(20), 5028. https://doi.org/10.3390/ijms20205028
[18]Doaei, S., Gholami, S., Rastgoo, S., Gholamalizadeh, M., Bourbour, F., Bagheri, S. E., Samipoor, F., Akbari, M. E., Shadnoush, M., Ghorat, F., Mosavi Jarrahi, S. A., Ashouri Mirsadeghi, N., Hajipour, A., Joola, P., Moslem, A., & Goodarzi, M. O. (2021). The effect of omega-3 fatty acid supplementation on clinical and biochemical parameters of critically ill patients with COVID-19: a randomized clinical trial. Journal of translational medicine, 19(1), 128. https://doi.org/10.1186/s12967-021-02795-5
[19]Mohammad, N. S., Nazli, R., Zafar, H., & Fatima, S. (2022). Effects of lipid based Multiple Micronutrients Supplement on the birth outcome of underweight pre-eclamptic women: A randomized clinical trial. Pakistan journal of medical sciences, 38(1), 219–226. https://doi.org/10.12669/pjms.38.1.4396
[20]Yang, C. P., Chang, C. M., Yang, C. C., Pariante, C. M., & Su, K. P. (2022). Long COVID and long chain fatty acids (LCFAs): Psychoneuroimmunity implication of omega-3 LCFAs in delayed consequences of COVID-19. Brain, behavior, and immunity, 103, 19–27. https://doi.org/10.1016/j.bbi.2022.04.001
[21]Kris-Etherton, P. M., Harris, W. S., Appel, L. J., & AHA Nutrition Committee. American Heart Association (2003). Omega-3 fatty acids and cardiovascular disease: new recommendations from the American Heart Association. Arteriosclerosis, thrombosis, and vascular biology, 23(2), 151–152. https://doi.org/10.1161/01.atv.0000057393.97337.ae
[22]Hathaway, D., Pandav, K., Patel, M., Riva-Moscoso, A., Singh, B. M., Patel, A., Min, Z. C., Singh-Makkar, S., Sana, M. K., Sanchez-Dopazo, R., Desir, R., Fahem, M. M. M., Manella, S., Rodriguez, I., Alvarez, A., & Abreu, R. (2020). Omega 3 Fatty Acids and COVID-19: A Comprehensive Review. Infection & chemotherapy, 52(4), 478–495. https://doi.org/10.3947/ic.2020.52.4.478
[23]McClaskey, E. M., & Michalets, E. L. (2007). Subdural hematoma after a fall in an elderly patient taking high-dose omega-3 fatty acids with warfarin and aspirin: case report and review of the literature. Pharmacotherapy, 27(1), 152–160. https://doi.org/10.1592/phco.27.1.152
[24]Shang, J., Smith, M. R., Anmangandla, A., & Lin, H. (2021). NAD+-consuming enzymes in immune defense against viral infection. Biochem J, 478(23), 4071-4092. doi:10.1042/BCJ20210181
[25]Jin, R., Niu, C., Wu, F., Zhou, S., Han, T., Zhang, Z., Li, E., Zhang, X., Xu, S., Wang, J., Tian, S., Chen, W., Ye, Q., Cao, C., & Cheng, L. (2022). DNA damage contributes to age-associated differences in SARS-CoV-2 infection. Aging cell, 21(12), e13729. https://doi.org/10.1111/acel.13729
[26]Elhassan, Y. S., Kluckova, K., Fletcher, R. S., Schmidt, M. S., Garten, A., Doig, C. L., Cartwright, D. M., Oakey, L., Burley, C. V., Jenkinson, N., Wilson, M., Lucas, S. J. E., Akerman, I., Seabright, A., Lai, Y. C., Tennant, D. A., Nightingale, P., Wallis, G. A., Manolopoulos, K. N., Brenner, C., … Lavery, G. G. (2019). Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-inflammatory Signatures. Cell reports, 28(7), 1717–1728.e6. https://doi.org/10.1016/j.celrep.2019.07.043
[27]Altay, O., Arif, M., Li, X., Yang, H., Aydın, M., Alkurt, G., Kim, W., Akyol, D., Zhang, C., Dinler-Doganay, G., Turkez, H., Shoaie, S., Nielsen, J., Borén, J., Olmuscelik, O., Doganay, L., Uhlén, M., & Mardinoglu, A. (2021). Combined Metabolic Activators Accelerates Recovery in Mild-to-Moderate COVID-19. Advanced science (Weinheim, Baden-Wurttemberg, Germany), 8(17), e2101222. https://doi.org/10.1002/advs.202101222
[28]Raines, N. H., Cheung, M. D., Wilson, L. S., Edberg, J. C., Erdmann, N. B., Schmaier, A. A., Berryhill, T. F., Manickas-Hill, Z., Li, J. Z., Yu, X. G., Agarwal, A., Barnes, S., & Parikh, S. M. (2021). Nicotinamide Adenine Dinucleotide Biosynthetic Impairment and Urinary Metabolomic Alterations Observed in Hospitalized Adults With COVID-19-Related Acute Kidney Injury. Kidney international reports, 6(12), 3002–3013. https://doi.org/10.1016/j.ekir.2021.09.001
[29]Jin, R., Niu, C., Wu, F., Zhou, S., Han, T., Zhang, Z., Li, E., Zhang, X., Xu, S., Wang, J., Tian, S., Chen, W., Ye, Q., Cao, C., & Cheng, L. (2022). DNA damage contributes to age-associated differences in SARS-CoV-2 infection. Aging cell, 21(12), e13729. https://doi.org/10.1111/acel.13729
[30]Babineau, T. J., Hackford, A., Kenler, A., Bistrian, B., Forse, R. A., Fairchild, P. G., Heard, S., Keroack, M., Caushaj, P., & Benotti, P. (1994). A phase II multicenter, double-blind, randomized, placebo-controlled study of three dosages of an immunomodulator (PGG-glucan) in high-risk surgical patients. Archives of surgery (Chicago, Ill. : 1960), 129(11), 1204–1210. https://doi.org/10.1001/archsurg.1994.01420350102014
[31]Sarinho, E., Medeiros, D., Schor, D., Rego Silva, A., Sales, V., Motta, M. E., Costa, A., Azoubel, A., & Rizzo, J. A. (2009). Production of interleukin-10 in asthmatic children after Beta-1-3-glucan. Allergologia et immunopathologia, 37(4), 188–192. https://doi.org/10.1016/j.aller.2009.02.005
[32]Lee, J. G., Kim, Y. S., Lee, Y. J., Ahn, H. Y., Kim, M., Kim, M., Cho, M. J., Cho, Y., & Lee, J. H. (2016). Effect of Immune-Enhancing Enteral Nutrition Enriched with or without Beta-Glucan on Immunomodulation in Critically Ill Patients. Nutrients, 8(6), 336. https://doi.org/10.3390/nu8060336
[33]Geller, A., & Yan, J. (2020). Could the Induction of Trained Immunity by β-Glucan Serve as a Defense Against COVID-19?. Frontiers in immunology, 11, 1782. https://doi.org/10.3389/fimmu.2020.01782
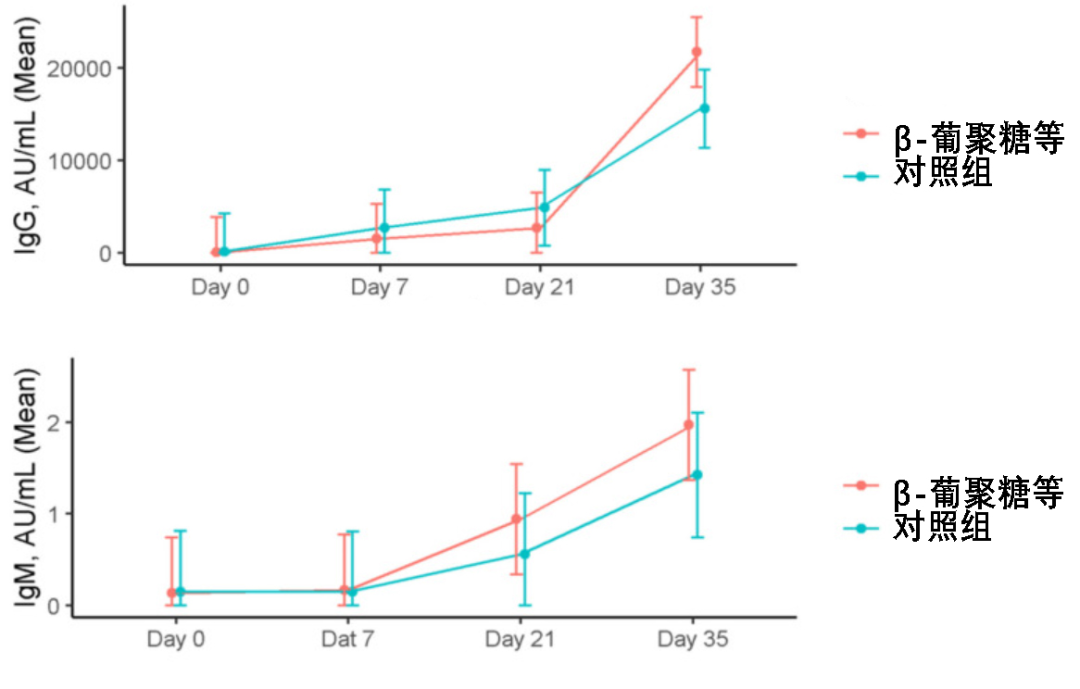
[34]Rodriguez, J. A. M., Bifano, M., Roca Goma, E., Plasencia, C. M., Torralba, A. O., Font, M. S., & Millán, P. R. (2021). Effect and Tolerability of a Nutritional Supplement Based on a Synergistic Combination of β-Glucans and Selenium- and Zinc-Enriched Saccharomyces cerevisiae (ABB C1®) in Volunteers Receiving the Influenza or the COVID-19 Vaccine: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients, 13(12), 4347. https://doi.org/10.3390/nu13124347
[35]Tanaka, A., Nishimura, M., Sato, Y., Sato, H., & Nishihira, J. (2015). Enhancement of the Th1-phenotype immune system by the intake of Oyster mushroom (Tamogitake) extract in a double-blind, placebo-controlled study. Journal of traditional and complementary medicine, 6(4), 424–430. https://doi.org/10.1016/j.jtcme.2015.11.004
[36]https://www.nature.com/articles/d42473-020-00414-3
[37]Di Pierro, F., Bertuccioli, A., & Cavecchia, I. (2020). Possible therapeutic role of a highly standardized mixture of active compounds derived from cultured Lentinula edodes mycelia (AHCC) in patients infected with 2019 novel coronavirus. Minerva gastroenterologica e dietologica, 66(2), 172–176. https://doi.org/10.23736/S1121-421X.20.02697-5
[38]Rayman M. P. (2012). Selenium and human health. Lancet (London, England), 379(9822), 1256–1268. https://doi.org/10.1016/S0140-6736(11)61452-9
[39]Zhang, J., Saad, R., Taylor, E. W., & Rayman, M. P. (2020). Selenium and selenoproteins in viral infection with potential relevance to COVID-19. Redox biology, 37, 101715. https://doi.org/10.1016/j.redox.2020.101715
[40]Mahmoodpoor, A., Hamishehkar, H., Shadvar, K., Ostadi, Z., Sanaie, S., Saghaleini, S. H., & Nader, N. D. (2019). The Effect of Intravenous Selenium on Oxidative Stress in Critically Ill Patients with Acute Respiratory Distress Syndrome. Immunological investigations, 48(2), 147–159. https://doi.org/10.1080/08820139.2018.1496098
[41]Broome, C. S., McArdle, F., Kyle, J. A., Andrews, F., Lowe, N. M., Hart, C. A., Arthur, J. R., & Jackson, M. J. (2004). An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. The American journal of clinical nutrition, 80(1), 154–162. https://doi.org/10.1093/ajcn/80.1.154
Vahedian-Azimi, A., Abbasifard, M., Rahimi-Bashar, F., Guest, P. C., Majeed, M., Mohammadi, A., Banach, M., Jamialahmadi, T., & Sahebkar, A. (2022). Effectiveness of Curcumin on Outcomes of Hospitalized COVID-19 Patients: A Systematic Review of Clinical Trials. Nutrients, 14(2), 256. https://doi.org/10.3390/nu14020256
[43]Kow, C. S., Ramachandram, D. S., & Hasan, S. S. (2022). The effect of curcumin on the risk of mortality in patients with COVID-19: A systematic review and meta-analysis of randomized trials. Phytotherapy research : PTR, 36(9), 3365–3368. https://doi.org/10.1002/ptr.7468
[44]Kumar Verma, A., Kumar, V., Singh, S., Goswami, B. C., Camps, I., Sekar, A., Yoon, S., & Lee, K. W. (2021). Repurposing potential of Ayurvedic medicinal plants derived active principles against SARS-CoV-2 associated target proteins revealed by molecular docking, molecular dynamics and MM-PBSA studies. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 137, 111356. https://doi.org/10.1016/j.biopha.2021.111356
[45]Rieder, A. S., Deniz, B. F., Netto, C. A., & Wyse, A. T. S. (2022). A Review of In Silico Research, SARS-CoV-2, and Neurodegeneration: Focus on Papain-Like Protease. Neurotoxicity research, 40(5), 1553–1569. https://doi.org/10.1007/s12640-022-00542-2
[46]Abdelazeem, B., Awad, A. K., Elbadawy, M. A., Manasrah, N., Malik, B., Yousaf, A., Alqasem, S., Banour, S., & Abdelmohsen, S. M. (2022). The effects of curcumin as dietary supplement for patients with COVID-19: A systematic review of randomized clinical trials. Drug discoveries & therapeutics, 16(1), 14–22. https://doi.org/10.5582/ddt.2022.01017
[47]Shoba, G., Joy, D., Joseph, T., Majeed, M., Rajendran, R., & Srinivas, P. S. (1998). Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta medica, 64(4), 353–356. https://doi.org/10.1055/s-2006-957450
[48]Lan, S. H., Lee, H. Z., Chao, C. M., Chang, S. P., Lu, L. C., & Lai, C. C. (2022). Efficacy of melatonin in the treatment of patients with COVID-19: A systematic review and meta-analysis of randomized controlled trials. Journal of medical virology, 94(5), 2102–2107. https://doi.org/10.1002/jmv.27595
[49]Molina-Carballo, A., Palacios-López, R., Jerez-Calero, A., Augustín-Morales, M. C., Agil, A., Muñoz-Hoyos, A., & Muñoz-Gallego, A. (2021). Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review. Current issues in molecular biology, 44(1), 31–45. https://doi.org/10.3390/cimb44010003
[50]Menczel Schrire, Z., Phillips, C. L., Chapman, J. L., Duffy, S. L., Wong, G., D'Rozario, A. L., Comas, M., Raisin, I., Saini, B., Gordon, C. J., McKinnon, A. C., Naismith, S. L., Marshall, N. S., Grunstein, R. R., & Hoyos, C. M. (2022). Safety of higher doses of melatonin in adults: A systematic review and meta-analysis. Journal of pineal research, 72(2), e12782. https://doi.org/10.1111/jpi.12782
[51]Di Pierro, F., Derosa, G., Maffioli, P., Bertuccioli, A., Togni, S., Riva, A., Allegrini, P., Khan, A., Khan, S., Khan, B. A., Altaf, N., Zahid, M., Ujjan, I. D., Nigar, R., Khushk, M. I., Phulpoto, M., Lail, A., Devrajani, B. R., & Ahmed, S. (2021). Possible Therapeutic Effects of Adjuvant Quercetin Supplementation Against Early-Stage COVID-19 Infection: A Prospective, Randomized, Controlled, and Open-Label Study. International journal of general medicine, 14, 2359–2366. https://doi.org/10.2147/IJGM.S318720
[52]Shohan, M., Nashibi, R., Mahmoudian-Sani, M. R., Abolnezhadian, F., Ghafourian, M., Alavi, S. M., Sharhani, A., & Khodadadi, A. (2022). The therapeutic efficacy of quercetin in combination with antiviral drugs in hospitalized COVID-19 patients: A randomized controlled trial. European journal of pharmacology, 914, 174615. https://doi.org/10.1016/j.ejphar.2021.174615
[53]Hemilä, H., Carr, A., & Chalker, E. (2021). Vitamin C May Increase the Recovery Rate of Outpatient Cases of SARS-CoV-2 Infection by 70%: Reanalysis of the COVID A to Z Randomized Clinical Trial. Frontiers in immunology, 12, 674681. https://doi.org/10.3389/fimmu.2021.674681
[54]Batista, K. S., Cintra, V. M., Lucena, P. A. F., Manhães-de-Castro, R., Toscano, A. E., Costa, L. P., Queiroz, M. E. B. S., de Andrade, S. M., Guzman-Quevedo, O., & Aquino, J. S. (2022). The role of vitamin B12 in viral infections: a comprehensive review of its relationship with the muscle-gut-brain axis and implications for SARS-CoV-2 infection. Nutrition reviews, 80(3), 561–578. https://doi.org/10.1093/nutrit/nuab092
[55]Tan, C. W., Ho, L. P., Kalimuddin, S., Cherng, B. P. Z., Teh, Y. E., Thien, S. Y., Wong, H. M., Tern, P. J. W., Chandran, M., Chay, J. W. M., Nagarajan, C., Sultana, R., Low, J. G. H., & Ng, H. J. (2020). Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition (Burbank, Los Angeles County, Calif.), 79-80, 111017. https://doi.org/10.1016/j.nut.2020.111017
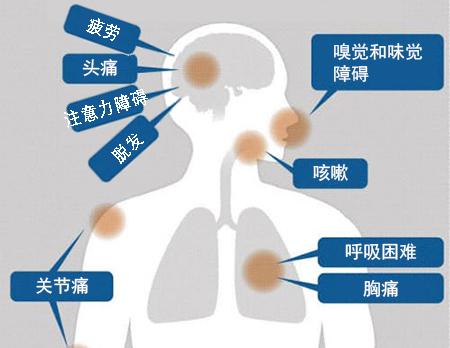
[56]Lopez-Leon, S., Wegman-Ostrosky, T., Perelman, C., Sepulveda, R., Rebolledo, P. A., Cuapio, A., & Villapol, S. (2021). More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. Scientific reports, 11(1), 16144. https://doi.org/10.1038/s41598-021-95565-8
[57]Dumont, R., Richard, V., Lorthe, E., Loizeau, A., Pennacchio, F., Zaballa, M. E., Baysson, H., Nehme, M., Perrin, A., L'Huillier, A. G., Kaiser, L., Barbe, R. P., Posfay-Barbe, K. M., Stringhini, S., SEROCoV-KIDS study group, & Guessous, I. (2022). A population-based serological study of post-COVID syndrome prevalence and risk factors in children and adolescents. Nature communications, 13(1), 7086. https://doi.org/10.1038/s41467-022-34616-8
[58]Salari, N., Khodayari, Y., Hosseinian-Far, A., Zarei, H., Rasoulpoor, S., Akbari, H., & Mohammadi, M. (2022). Global prevalence of chronic fatigue syndrome among long COVID-19 patients: A systematic review and meta-analysis. BioPsychoSocial medicine, 16(1), 21. https://doi.org/10.1186/s13030-022-00250-5
[59]Ramani, S. L., Samet, J., Franz, C. K., Hsieh, C., Nguyen, C. V., Horbinski, C., & Deshmukh, S. (2021). Musculoskeletal involvement of COVID-19: review of imaging. Skeletal radiology, 50(9), 1763–1773. https://doi.org/10.1007/s00256-021-03734-7
[60]Tuominen, L. J., Sokolowski, S., Lundell, R. V., & Räisänen-Sokolowski, A. K. (2022). Decompression illness in Finnish technical divers: a follow-up study on incidence and self-treatment. Diving and hyperbaric medicine, 52(2), 78–84. https://doi.org/10.28920/dhm52.2.74-84
[61]Kjellberg, A., Abdel-Halim, L., Hassler, A., El Gharbi, S., Al-Ezerjawi, S., Boström, E., Sundberg, C. J., Pernow, J., Medson, K., Kowalski, J. H., Rodriguez-Wallberg, K. A., Zheng, X., Catrina, S., Runold, M., Ståhlberg, M., Bruchfeld, J., Nygren-Bonnier, M., & Lindholm, P. (2022). Hyperbaric oxygen for treatment of long COVID-19 syndrome (HOT-LoCO): protocol for a randomised, placebo-controlled, double-blind, phase II clinical trial. BMJ open, 12(11), e061870. https://doi.org/10.1136/bmjopen-2022-061870
[62]Tibullo, D., Li Volti, G., Giallongo, C., Grasso, S., Tomassoni, D., Anfuso, C. D., Lupo, G., Amenta, F., Avola, R., & Bramanti, V. (2017). Biochemical and clinical relevance of alpha lipoic acid: antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflammation research : official journal of the European Histamine Research Society ... [et al.], 66(11), 947–959. https://doi.org/10.1007/s00011-017-1079-6
[63]Rahimlou, M., Asadi, M., Banaei Jahromi, N., & Mansoori, A. (2019). Alpha-lipoic acid (ALA) supplementation effect on glycemic and inflammatory biomarkers: A Systematic Review and meta- analysis. Clinical nutrition ESPEN, 32, 16–28. https://doi.org/10.1016/j.clnesp.2019.03.015
[64]Hidaka, T., Fujii, K., Funahashi, I., Fukutomi, N., & Hosoe, K. (2008). Safety assessment of coenzyme Q10 (CoQ10). BioFactors (Oxford, England), 32(1-4), 199–208. https://doi.org/10.1002/biof.5520320124
[65]Barletta, M. A., Marino, G., Spagnolo, B., Bianchi, F. P., Falappone, P. C. F., Spagnolo, L., & Gatti, P. (2022). Coenzyme Q10 + alpha lipoic acid for chronic COVID syndrome. Clinical and experimental medicine, 1–12. Advance online publication. https://doi.org/10.1007/s10238-022-00871-8
[66]Castro-Marrero, J., Segundo, M. J., Lacasa, M., Martinez-Martinez, A., Sentañes, R. S., & Alegre-Martin, J. (2021). Effect of Dietary Coenzyme Q10 Plus NADH Supplementation on Fatigue Perception and Health-Related Quality of Life in Individuals with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Prospective, Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients, 13(8), 2658. https://doi.org/10.3390/nu13082658
[67]Castro-Marrero, J., Domingo, J. C., Cordobilla, B., Ferrer, R., Giralt, M., Sanmartín-Sentañes, R., & Alegre-Martín, J. (2022). Does Coenzyme Q10 Plus Selenium Supplementation Ameliorate Clinical Outcomes by Modulating Oxidative Stress and Inflammation in Individuals with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome?. Antioxidants & redox signaling, 36(10-12), 729–739. https://doi.org/10.1089/ars.2022.0018
[68]Teitelbaum, J. E., Johnson, C., & St Cyr, J. (2006). The use of D-ribose in chronic fatigue syndrome and fibromyalgia: a pilot study. Journal of alternative and complementary medicine (New York, N.Y.), 12(9), 857–862. https://doi.org/10.1089/acm.2006.12.857
[69]Tosato, M., Calvani, R., Picca, A., Ciciarello, F., Galluzzo, V., Coelho-Júnior, H. J., Di Giorgio, A., Di Mario, C., Gervasoni, J., Gremese, E., Leone, P. M., Nesci, A., Paglionico, A. M., Santoliquido, A., Santoro, L., Santucci, L., Tolusso, B., Urbani, A., Marini, F., Marzetti, E., … Gemelli against COVID-19 Post-Acute Care Team (2022). Effects of l-Arginine Plus Vitamin C Supplementation on Physical Performance, Endothelial Function, and Persistent Fatigue in Adults with Long COVID: A Single-Blind Randomized Controlled Trial. Nutrients, 14(23), 4984. https://doi.org/10.3390/nu14234984
[70]Regland, B., Forsmark, S., Halaouate, L., Matousek, M., Peilot, B., Zachrisson, O., & Gottfries, C. G. (2015). Response to vitamin B12 and folic acid in myalgic encephalomyelitis and fibromyalgia. PloS one, 10(4), e0124648. https://doi.org/10.1371/journal.pone.0124648
[71]Askari, G., Sahebkar, A., Soleimani, D., Mahdavi, A., Rafiee, S., Majeed, M., Khorvash, F., Iraj, B., Elyasi, M., Rouhani, M. H., & Bagherniya, M. (2022). The efficacy of curcumin-piperine co-supplementation on clinical symptoms, duration, severity, and inflammatory factors in COVID-19 outpatients: a randomized double-blind, placebo-controlled trial. Trials, 23(1), 472. https://doi.org/10.1186/s13063-022-06375-w
[72]Shauer, A., Gotsman, I., Keren, A., Zwas, D. R., Hellman, Y., Durst, R., & Admon, D. (2013). Acute viral myocarditis: current concepts in diagnosis and treatment. The Israel Medical Association journal : IMAJ, 15(3), 180–185.
[73]Cooper L. T., Jr (2009). Myocarditis. The New England journal of medicine, 360(15), 1526–1538. https://doi.org/10.1056/NEJMra0800028
[74]Yin, Y. J., Zeng, S. L., Li, Y. W., Wu, Z., Huang, D. J., & Tang, H. Z. (2021). The effect of coenzyme Q10 plus trimetazidine on acute viral myocarditis treatment. American journal of translational research, 13(12), 13854–13861.
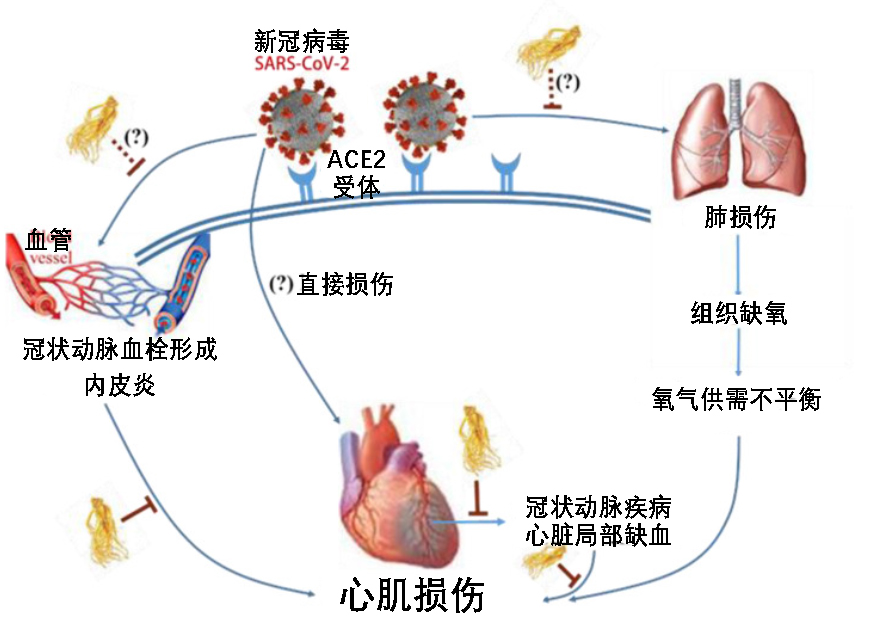
[75]Hossain, M. A., & Kim, J. H. (2022). Possibility as role of ginseng and ginsenosides on inhibiting the heart disease of COVID-19: A systematic review. Journal of ginseng research, 46(3), 321–330. https://doi.org/10.1016/j.jgr.2022.01.003
[76]Ji, S., Li, R., Wang, Q., Miao, W. J., Li, Z. W., Si, L. L., Qiao, X., Yu, S. W., Zhou, D. M., & Ye, M. (2015). Anti-H1N1 virus, cytotoxic and Nrf2 activation activities of chemical constituents from Scutellaria baicalensis. Journal of ethnopharmacology, 176, 475–484. https://doi.org/10.1016/j.jep.2015.11.018
[77]Oo, A., Teoh, B. T., Sam, S. S., Bakar, S. A., & Zandi, K. (2019). Baicalein and baicalin as Zika virus inhibitors. Archives of virology, 164(2), 585–593. https://doi.org/10.1007/s00705-018-4083-4
[78]Zandi, K., Lim, T. H., Rahim, N. A., Shu, M. H., Teoh, B. T., Sam, S. S., Danlami, M. B., Tan, K. K., & Abubakar, S. (2013). Extract of Scutellaria baicalensis inhibits dengue virus replication. BMC complementary and alternative medicine, 13, 91. https://doi.org/10.1186/1472-6882-13-91
[79]Song, J., Zhang, L., Xu, Y., Yang, D., Zhang, L., Yang, S., Zhang, W., Wang, J., Tian, S., Yang, S., Yuan, T., Liu, A., Lv, Q., Li, F., Liu, H., Hou, B., Peng, X., Lu, Y., & Du, G. (2021). The comprehensive study on the therapeutic effects of baicalein for the treatment of COVID-19 in vivo and in vitro. Biochemical pharmacology, 183, 114302. https://doi.org/10.1016/j.bcp.2020.114302
[80]Chen, L., & Zhao, W. (2016). Apigenin protects against bleomycin-induced lung fibrosis in rats. Experimental and therapeutic medicine, 11(1), 230–234. https://doi.org/10.3892/etm.2015.2885
[81]Cardenas, H., Arango, D., Nicholas, C., Duarte, S., Nuovo, G. J., He, W., Voss, O. H., Gonzalez-Mejia, M. E., Guttridge, D. C., Grotewold, E., & Doseff, A. I. (2016). Dietary Apigenin Exerts Immune-Regulatory Activity in Vivo by Reducing NF-κB Activity, Halting Leukocyte Infiltration and Restoring Normal Metabolic Function. International journal of molecular sciences, 17(3), 323. https://doi.org/10.3390/ijms17030323
[82]Pang, L., Zou, S., Shi, Y., Mao, Q., & Chen, Y. (2019). Apigenin attenuates PM2.5-induced airway hyperresponsiveness and inflammation by down-regulating NF-κB in murine model of asthma. International journal of clinical and experimental pathology, 12(10), 3700–3709.
[83]de Alencar, J. C. G., Moreira, C. L., Müller, A. D., Chaves, C. E., Fukuhara, M. A., da Silva, E. A., Miyamoto, M. F. S., Pinto, V. B., Bueno, C. G., Lazar Neto, F., Gomez Gomez, L. M., Menezes, M. C. S., Marchini, J. F. M., Marino, L. O., Brandão Neto, R. A., Souza, H. P., & COVID Register Group (2021). Double-blind, Randomized, Placebo-controlled Trial With N-acetylcysteine for Treatment of Severe Acute Respiratory Syndrome Caused by Coronavirus Disease 2019 (COVID-19). Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 72(11), e736–e741. https://doi.org/10.1093/cid/ciaa1443
[84]Tenório, M. C. D. S., Graciliano, N. G., Moura, F. A., Oliveira, A. C. M., & Goulart, M. O. F. (2021). N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants (Basel, Switzerland), 10(6), 967. https://doi.org/10.3390/antiox10060967
[85]Chen, J., Liu, D., Liu, L., Liu, P., Xu, Q., Xia, L., Ling, Y., Huang, D., Song, S., Zhang, D., Qian, Z., Li, T., Shen, Y., & Lu, H. (2020). Zhejiang da xue xue bao. Yi xue ban = Journal of Zhejiang University. Medical sciences, 49(2), 215–219. https://doi.org/10.3785/j.issn.1008-9292.2020.03.03
[86]Firoz, A., & Talwar, P. (2022). COVID-19 and retinal degenerative diseases: Promising link "Kaempferol". Current opinion in pharmacology, 64, 102231. https://doi.org/10.1016/j.coph.2022.102231
[87]Gattinoni, L., Chiumello, D., Caironi, P., Busana, M., Romitti, F., Brazzi, L., & Camporota, L. (2020). COVID-19 pneumonia: different respiratory treatments for different phenotypes?. Intensive care medicine, 46(6), 1099–1102. https://doi.org/10.1007/s00134-020-06033-2
[88]Sokolovska, L., Terentjeva-Decuka, A., Cistjakovs, M., Nora-Krukle, Z., Gravelsina, S., Vilmane, A., Vecvagare, K., & Murovska, M. (2022). The presence of SARS-CoV-2 in multiple clinical specimens of a fatal case of COVID-19: a case report. Journal of medical case reports, 16(1), 484. https://doi.org/10.1186/s13256-022-03706-y
[89]Xu, L., Hu, Z., Shen, J., & McQuillan, P. M. (2015). Effects of Ginkgo biloba extract on cerebral oxygen and glucose metabolism in elderly patients with pre-existing cerebral ischemia. Complementary therapies in medicine, 23(2), 220–225. https://doi.org/10.1016/j.ctim.2014.12.009
[90]Lu, Y., Li, X., Geng, D., Mei, N., Wu, P. Y., Huang, C. C., Jia, T., Zhao, Y., Wang, D., Xiao, A., & Yin, B. (2020). Cerebral Micro-Structural Changes in COVID-19 Patients - An MRI-based 3-month Follow-up Study. EClinicalMedicine, 25, 100484. https://doi.org/10.1016/j.eclinm.2020.100484
[91]Taquet, M., Geddes, J. R., Husain, M., Luciano, S., & Harrison, P. J. (2021). 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. The lancet. Psychiatry, 8(5), 416–427. https://doi.org/10.1016/S2215-0366(21)00084-5
[92]Zhao, S., Shibata, K., Hellyer, P. J., Trender, W., Manohar, S., Hampshire, A., & Husain, M. (2022). Rapid vigilance and episodic memory decrements in COVID-19 survivors. Brain communications, 4(1), fcab295. https://doi.org/10.1093/braincomms/fcab295
[93]De Luca, P., Camaioni, A., Marra, P., Salzano, G., Carriere, G., Ricciardi, L., Pucci, R., Montemurro, N., Brenner, M. J., & Di Stadio, A. (2022). Effect of Ultra-Micronized Palmitoylethanolamide and Luteolin on Olfaction and Memory in Patients with Long COVID: Results of a Longitudinal Study. Cells, 11(16), 2552. https://doi.org/10.3390/cells11162552
[94]Di Stadio, A., D'Ascanio, L., Vaira, L. A., Cantone, E., De Luca, P., Cingolani, C., Motta, G., De Riu, G., Vitelli, F., Spriano, G., De Vincentiis, M., Camaioni, A., La Mantia, I., Ferreli, F., & Brenner, M. J. (2022). Ultramicronized Palmitoylethanolamide and Luteolin Supplement Combined with Olfactory Training to Treat Post-COVID-19 Olfactory Impairment: A Multi-Center Double-Blinded Randomized Placebo- Controlled Clinical Trial. Current neuropharmacology, 20(10), 2001–2012. https://doi.org/10.2174/1570159X20666220420113513
[95]D'Ascanio, L., Vitelli, F., Cingolani, C., Maranzano, M., Brenner, M. J., & Di Stadio, A. (2021). Randomized clinical trial "olfactory dysfunction after COVID-19: olfactory rehabilitation therapy vs. intervention treatment with Palmitoylethanolamide and Luteolin": preliminary results. European review for medical and pharmacological sciences, 25(11), 4156–4162. https://doi.org/10.26355/eurrev_202106_26059
[96]Di Stadio, A., & Angelini, C. (2018). Microglia polarization by mitochondrial metabolism modulation: A therapeutic opportunity in neurodegenerative diseases. Mitochondrion, 46, 334-336.
[97]Campolo, M., Crupi, R., Cordaro, M., Cardali, S. M., Ardizzone, A., Casili, G., ... & Cuzzocrea, S. (2021). Co-ultra PEALut enhances endogenous repair response following moderate traumatic brain injury. International Journal of Molecular Sciences, 22(16), 8717.
Bartlett, J. A., & van der Voort Maarschalk, K. (2012). Understanding the oral mucosal absorption and resulting clinical pharmacokinetics of asenapine. AAPS PharmSciTech, 13(4), 1110–1115. https://doi.org/10.1208/s12249-012-9839-7