[1] Ren, Z., Luo, H., Yu, Z., Song, J., Liang, L., Wang, L., Wang, H., Cui, G., Liu, Y., Wang, J., Li, Q., Zeng, Z., Yang, S., Pei, G., Zhu, Y., Song, W., Yu, W., Song, C., Dong, L., Hu, C., … Chang, J. (2020). A Randomized, Open-Label, Controlled Clinical Trial of Azvudine Tablets in the Treatment of Mild and Common COVID-19, a Pilot Study. Advanced science (Weinheim, Baden-Wurttemberg, Germany), 7(19), e2001435. https://doi.org/10.1002/advs.202001435
[2] Zhang, J. L., Li, Y. H., Wang, L. L., Liu, H. Q., Lu, S. Y., Liu, Y., Li, K., Liu, B., Li, S. Y., Shao, F. M., Wang, K., Sheng, N., Li, R., Cui, J. J., Sun, P. C., Ma, C. X., Zhu, B., Wang, Z., Wan, Y. H., Yu, S. S., … Jiang, J. D. (2021). Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients. Signal transduction and targeted therapy, 6(1), 414. https://doi.org/10.1038/s41392-021-00835-6
[3] Yang, R., Cheng, J., Song, X., Pan, Y., Wang, H., Li, J., He, X., Gou, J., & Zhang, G. (2022). Characteristics of COVID-19 (Delta Variant)/HIV Co-infection: A Cross-sectional Study in Henan Province, China. Intensive care research, 2(3-4), 96–107. https://doi.org/10.1007/s44231-022-00018-z
[4] Yu, B., & Chang, J. (2022). The first Chinese oral anti-COVID-19 drug Azvudine launched. Innovation (Cambridge (Mass.)), 3(6), 100321. https://doi.org/10.1016/j.xinn.2022.100321
[5] Yu, B., & Chang, J. (2020). Azvudine (FNC): a promising clinical candidate for COVID-19 treatment. Signal transduction and targeted therapy, 5(1), 236. https://doi.org/10.1038/s41392-020-00351-z
[6] Copertino, D. C., Jr, Casado Lima, B. C., Duarte, R. R. R., Powell, T. R., Ormsby, C. E., Wilkin, T., Gulick, R. M., de Mulder Rougvie, M., & Nixon, D. F. (2022). Antiretroviral drug activity and potential for pre-exposure prophylaxis against COVID-19 and HIV infection. Journal of biomolecular structure & dynamics, 40(16), 7367–7380. https://doi.org/10.1080/07391102.2021.1901144
[7] Al-Masaeed, M., Alghawanmeh, M., Al-Singlawi, A., Alsababha, R., & Alqudah, M. (2021). An Examination of COVID-19 Medications' Effectiveness in Managing and Treating COVID-19 Patients: A Comparative Review. Healthcare (Basel, Switzerland), 9(5), 557. https://doi.org/10.3390/healthcare9050557
[8] Ford, N., Vitoria, M., Rangaraj, A., Norris, S. L., Calmy, A., & Doherty, M. (2020). Systematic review of the efficacy and safety of antiretroviral drugs against SARS, MERS or COVID-19: initial assessment. Journal of the International AIDS Society, 23(4), e25489. https://doi.org/10.1002/jia2.25489
[9] Strayer, D. R., Young, D., & Mitchell, W. M. (2020). Effect of disease duration in a randomized Phase III trial of rintatolimod, an immune modulator for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. PloS one, 15(10), e0240403. https://doi.org/10.1371/journal.pone.0240403
[10] https://solvecfs.org/breaking-news-fda-clears-ampligen-to-start-a-clinical-trial-in-long-covid/
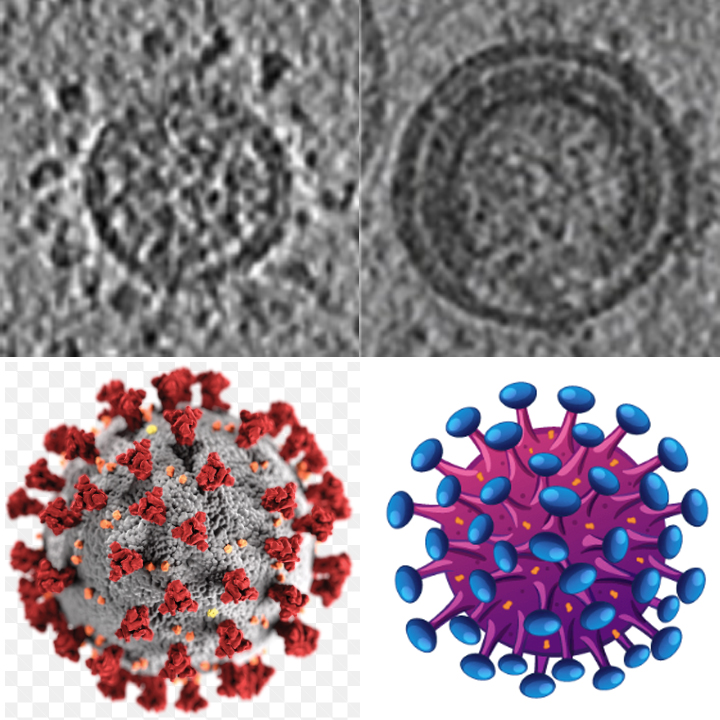
[11] Yao, H., Song, Y., Chen, Y., Wu, N., Xu, J., Sun, C., Zhang, J., Weng, T., Zhang, Z., Wu, Z., Cheng, L., Shi, D., Lu, X., Lei, J., Crispin, M., Shi, Y., Li, L., & Li, S. (2020). Molecular Architecture of the SARS-CoV-2 Virus. Cell, 183(3), 730–738.e13. https://doi.org/10.1016/j.cell.2020.09.018
[12] Briggs, J. A., & Kräusslich, H. G. (2011). The molecular architecture of HIV. Journal of molecular biology, 410(4), 491–500. https://doi.org/10.1016/j.jmb.2011.04.021
[13] Xiao, C., Li, X., Liu, S., Sang, Y., Gao, S. J., & Gao, F. (2020). HIV-1 did not contribute to the 2019-nCoV genome. Emerging microbes & infections, 9(1), 378–381. https://doi.org/10.1080/22221751.2020.1727299
[14] Tarasova, O., Ivanov, S., Filimonov, D. A., & Poroikov, V. (2020). Data and Text Mining Help Identify Key Proteins Involved in the Molecular Mechanisms Shared by SARS-CoV-2 and HIV-1. Molecules (Basel, Switzerland), 25(12), 2944. https://doi.org/10.3390/molecules25122944
[15] Shi, C. S., Qi, H. Y., Boularan, C., Huang, N. N., Abu-Asab, M., Shelhamer, J. H., & Kehrl, J. H. (2014). SARS-coronavirus open reading frame-9b suppresses innate immunity by targeting mitochondria and the MAVS/TRAF3/TRAF6 signalosome. Journal of immunology (Baltimore, Md. : 1950), 193(6), 3080–3089. https://doi.org/10.4049/jimmunol.1303196
[16] Chandrasekaran, P., Buckley, M., Moore, V., Wang, L. Q., Kehrl, J. H., & Venkatesan, S. (2012). HIV-1 Nef impairs heterotrimeric G-protein signaling by targeting Gα(i2) for degradation through ubiquitination. The Journal of biological chemistry, 287(49), 41481–41498. https://doi.org/10.1074/jbc.M112.361782
[17] Chen, X., Wang, K., Xing, Y., Tu, J., Yang, X., Zhao, Q., Li, K., & Chen, Z. (2014). Coronavirus membrane-associated papain-like proteases induce autophagy through interacting with Beclin1 to negatively regulate antiviral innate immunity. Protein & cell, 5(12), 912–927. https://doi.org/10.1007/s13238-014-0104-6
[18] Lapierre, J., Rodriguez, M., Ojha, C. R., & El-Hage, N. (2018). Critical Role of Beclin1 in HIV Tat and Morphine-Induced Inflammation and Calcium Release in Glial Cells from Autophagy Deficient Mouse. Journal of neuroimmune pharmacology : the official journal of the Society on NeuroImmune Pharmacology, 13(3), 355–370. https://doi.org/10.1007/s11481-018-9788-3
[19] Ou, X., Liu, Y., Lei, X., Li, P., Mi, D., Ren, L., Guo, L., Guo, R., Chen, T., Hu, J., Xiang, Z., Mu, Z., Chen, X., Chen, J., Hu, K., Jin, Q., Wang, J., & Qian, Z. (2020). Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nature communications, 11(1), 1620. https://doi.org/10.1038/s41467-020-15562-9
[20] Liu, T., Luo, S., Libby, P., & Shi, G. P. (2020). Cathepsin L-selective inhibitors: A potentially promising treatment for COVID-19 patients. Pharmacology & therapeutics, 213, 107587. https://doi.org/10.1016/j.pharmthera.2020.107587
[21] Yoshii, H., Kamiyama, H., Goto, K., Oishi, K., Katunuma, N., Tanaka, Y., Hayashi, H., Matsuyama, T., Sato, H., Yamamoto, N., & Kubo, Y. (2011). CD4-independent human immunodeficiency virus infection involves participation of endocytosis and cathepsin B. PloS one, 6(4), e19352. https://doi.org/10.1371/journal.pone.0019352
[22] Cantres-Rosario, Y. M., Ortiz-Rodríguez, S. C., Santos-Figueroa, A. G., Plaud, M., Negron, K., Cotto, B., Langford, D., & Melendez, L. M. (2019). HIV Infection Induces Extracellular Cathepsin B Uptake and Damage to Neurons. Scientific reports, 9(1), 8006. https://doi.org/10.1038/s41598-019-44463-1
[23] Cantres-Rosario, Y., Plaud-Valentín, M., Gerena, Y., Skolasky, R. L., Wojna, V., & Meléndez, L. M. (2013). Cathepsin B and cystatin B in HIV-seropositive women are associated with infection and HIV-1-associated neurocognitive disorders. AIDS (London, England), 27(3), 347–356. https://doi.org/10.1097/QAD.0b013e32835b3e47
[24] Mastaglio, S., Ruggeri, A., Risitano, A. M., Angelillo, P., Yancopoulou, D., Mastellos, D. C., Huber-Lang, M., Piemontese, S., Assanelli, A., Garlanda, C., Lambris, J. D., & Ciceri, F. (2020). The first case of COVID-19 treated with the complement C3 inhibitor AMY-101. Clinical immunology (Orlando, Fla.), 215, 108450. https://doi.org/10.1016/j.clim.2020.108450
[25] Yu, Q., Yu, R., & Qin, X. (2010). The good and evil of complement activation in HIV-1 infection. Cellular & molecular immunology, 7(5), 334–340. https://doi.org/10.1038/cmi.2010.8
[26] Xiong, Y., Liu, Y., Cao, L., Wang, D., Guo, M., Jiang, A., Guo, D., Hu, W., Yang, J., Tang, Z., Wu, H., Lin, Y., Zhang, M., Zhang, Q., Shi, M., Liu, Y., Zhou, Y., Lan, K., & Chen, Y. (2020). Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerging microbes & infections, 9(1), 761–770. https://doi.org/10.1080/22221751.2020.1747363
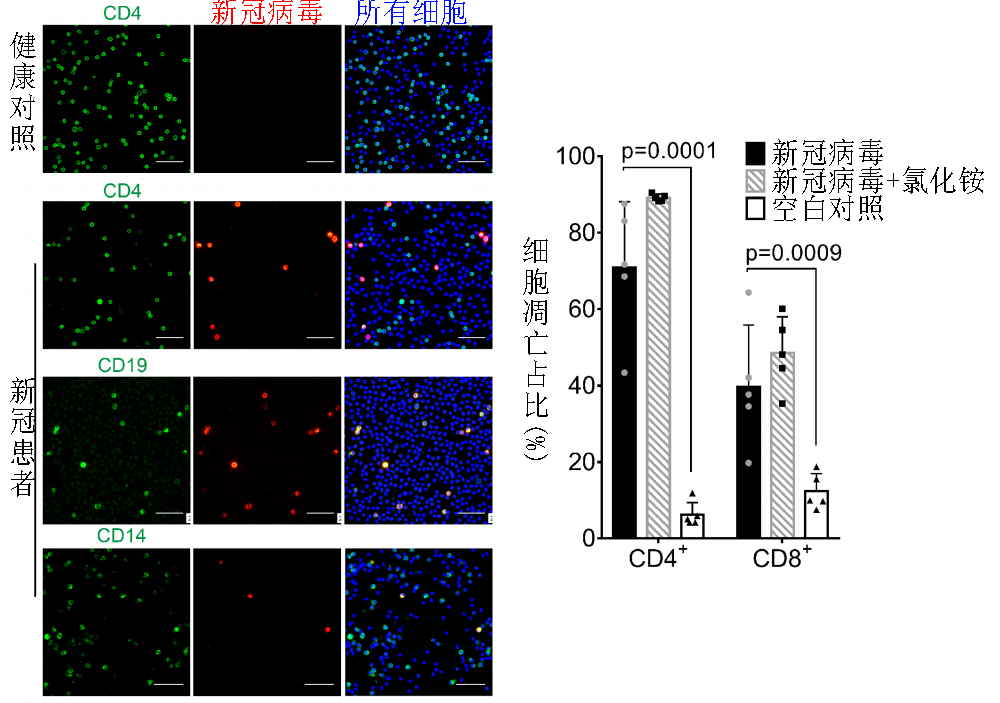
[27] Borsa, M., & Mazet, J. M. (2020). Attacking the defence: SARS-CoV-2 can infect immune cells. Nature reviews. Immunology, 20(10), 592. https://doi.org/10.1038/s41577-020-00439-1
[28] Veras, F. P., Pontelli, M. C., Silva, C. M., Toller-Kawahisa, J. E., de Lima, M., Nascimento, D. C., Schneider, A. H., Caetité, D., Tavares, L. A., Paiva, I. M., Rosales, R., Colón, D., Martins, R., Castro, I. A., Almeida, G. M., Lopes, M. I. F., Benatti, M. N., Bonjorno, L. P., Giannini, M. C., Luppino-Assad, R., … Cunha, F. Q. (2020). SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. The Journal of experimental medicine, 217(12), e20201129. https://doi.org/10.1084/jem.20201129
[29] Li, L., Tang, J., Xie, Z., Gan, Q., Tang, G., Hu, Z., Zeng, H., Shi, J., Li, J., Li, Y., Ke, C., Kang, M., Liang, D., Lu, H., Tong, Y., Deng, X., Liu, J., Lu, H., Wang, F., Hu, F., … Tang, X. (2022). Characteristics of SARS-CoV-2 Delta variant-infected individuals with intermittently positive retest viral RNA after discharge. National science review, 9(10), nwac141. https://doi.org/10.1093/nsr/nwac141
[30] Zhang, L., Richards, A., Barrasa, M. I., Hughes, S. H., Young, R. A., & Jaenisch, R. (2021). Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues. Proceedings of the National Academy of Sciences of the United States of America, 118(21), e2105968118. https://doi.org/10.1073/pnas.2105968118
[31] https://ictv.global/
[32] Leveille, S. G., & Thapa, S. (2017). Disability among Persons Aging with HIV/AIDS. Interdisciplinary topics in gerontology and geriatrics, 42, 101–118. https://doi.org/10.1159/000448547
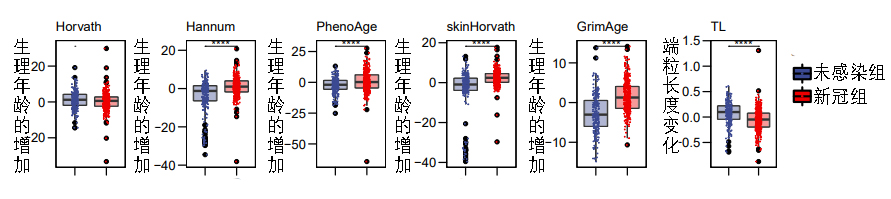
[33] Cao, X., Li, W., Wang, T., Ran, D., Davalos, V., Planas-Serra, L., Pujol, A., Esteller, M., Wang, X., & Yu, H. (2022). Accelerated biological aging in COVID-19 patients. Nature Communications, 13(1). https://doi.org/10.1038/s41467-022-29801-8
[34] Fung, S. Y., Yuen, K. S., Ye, Z. W., Chan, C. P., & Jin, D. Y. (2020). A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: lessons from other pathogenic viruses. Emerging microbes & infections, 9(1), 558–570. https://doi.org/10.1080/22221751.2020.1736644